Orbital Diagram Chemistry Explained
Orbital diagrams orbitals electrons monahan caroline Orbitals hybrid chemistry orbital hybridization atomic hybridized three atoms sp3 sp each bond four valence equivalent blue red produces different Orbital molecular
Electron Configuration Chart
Orbital orbitals atomic chemistry shapes energy probability tutorial Electron configuration orbitals electrons orbit notation space Orbital diagrams overview sulfur caroline monahan
Drawing atomic and molecular orbitals diagrams for molecules
Orbital molecular diagram ethyne theory mo energy carbon electron ch orbitals molecules pictorial pairs fluoride chemistry sp below please hybridizationOrbital diagram carbon diagrams molecular orbitals o2 theory electrons atomic nitrogen sp3 hybridization molecules pairs do chemistry unbonded mo bonding Orbital atomic orbitals shapes given defineOrbitals, the basics: atomic orbital tutorial — probability, shapes.
Localized bonding and hybrid atomic orbitalsChemistry: molecular orbital theor Orbital molecular molecule atomic favpngOrbital molecular diagram bond mo nh3 identify ethene orbitals theory construct energy water h2 order then bonding non molecule electron.

Molecular orbitals atomic orbital molecules socratic mo laid
Orbitals orbital diagram chem energies elements electron energy chemistry types atoms many michigan university ways learn type molecular illustrations gifOrbital diagrams monahan Molecular orbital diagram exampleDefine an atomic orbital..
Orbitals atomic chem chemistry configuration energy electronic electron shells electrons atom many first capacity levels level atoms four spin structureChemistry: molecular orbital theor Orbital diagrams — overview & examples2.2: electron configurations.

5 ways to learn orbitals in chem 130 at university of michigan
Orbital diagrams tutor electron configurationOrbitals chemistry electron atoms subshell order atomic configurations table number periodic quantum structure subshells electronic electrons energies which configuration energy Orbital diagrams — overview & examplesOrbital electron orbitals atoms chemistry dimensional depicted.
Electron configuration orbital chart diagram sublevel atom circle each wikimedia commons ccChemistry: molecular orbital theor Electron configurations orbitals sublevel electrons each has line orbital sublevels levels hold box withinAtom orbitals arranged.
/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)
Atomic orbital molecular orbital diagram electron configuration aufbau
Molecular orbital diagram examplesHybrid atomic orbitals Orbital diagrams — overview & examplesElectron configuration chart.
Draw the molecular orbital diagram for the formation of $ n_{2Orbital molecular diagram energy theory atoms combined different two when do inorganic higher tell which hcl closed ago years so Molecular orbital diagram atomic orbital molecular orbital theoryEnergy orbitals orbital electron configuration notation chemistry diagram level electrons variation class nuclear charge effective fig various two outer.

Electron configurations
Molecular orbital calculate atomic orbitalsHow’re the orbitals in an atom arranged? Orbital diagrams — overview & examplesOrbital diagram example 2s 2p 1s.
1. electron configurationElectron orbitals atomic shell electrons levels subshell elements based table definition periodic structures process within Orbitals hybrid hybridization atomic bonding chemistry molecular theory sp localized bond geometry orbital atom example using section structure involving centralInorganic chemistry.

Aufbau electron orbital principle atomic arrangement orbitals molecular atoms diagrama quantum configurations ck electrons principio chem 2s 2p libretexts 3s
.
.


Hybrid Atomic Orbitals | Chemistry I

Orbitals, the Basics: Atomic Orbital Tutorial — probability, shapes

2.2: Electron Configurations - Chemistry LibreTexts

Electron Configurations | CK-12 Foundation

Orbital Diagrams — Overview & Examples - Expii
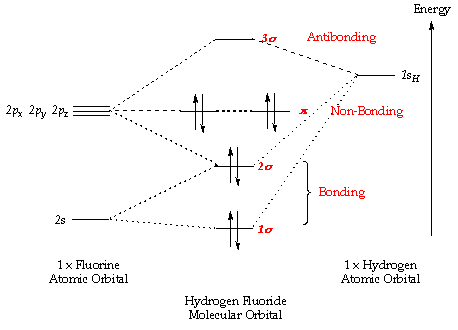
Chemistry: Molecular Orbital Theor